An Investigation into the change in rate of reaction of the catalase enzyme based on changes in pH11/7/2016 ObjectiveDuring this experiment we wanted to see the affect of different pH levels on the rate of reaction on the enzyme catalase. Catalase is an enzyme found commonly in every cell in our bodies and is vital to our survival. It converts the toxic bi-product of respiration, Hydrogen peroxide, to water and oxygen. HypothesisI predict that the enzyme will function the most efficiently in the alkaline environment. Most likely at a 9-10 pH as it has to function inside the cell cytoplasm ApparatusSafety Goggles Timer Spotting Tile Petri dish of filter paper discs forceps 10ml syringe (for distilled water) 2ml syringe (for Hydrogen Peroxide) 1ml Pipette (vol of bean juice! & Buffer solution) 10 mung beans (catalase!) Hydrogen peroxide (1% concentration) Distilled water pH buffers Washable marker pen Peroxide containers Method
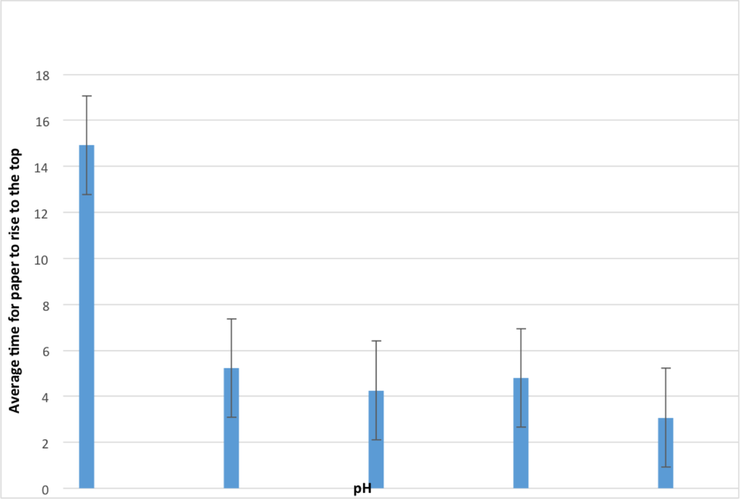
ResultsWhat does this evidence show? Clearly we can see a huge decrease in the length of time required to send the paper to the top, when the pH increases (note the pH increases from right to left going 5,7,8,9,10). What this shows is that the best conditions for this enzyme to work in is an alkaline condition. We can also safely assume that the optimum pH is 10 as this is the one with the lowest average time to produce enough oxygen to float the disc. However there is no significant impact on the results from the alkaline as the error bars overlap. However with our acidic result we see no overlap. So this suggests there may be a significant difference in the results. This is supported by the fact that the enzyme would have been denatured, and so would have had a significant impact on the rate of reaction. So I can conclude my hypothesis correct. How to Improve the ExperimentThe main aspect about the experiment that could of been improved, is the number of tests on the lower pH. We should have included more acidic pH's, in order to confirm the exact pH where the enzyme denatures. We could of also had more repeats to increase reliability as the results are quite close.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
